Blockchain for Supply Chain in Pharma, Healthcare & Life Sciences — Detailed Explanation
Blockchain in pharmaceutical and healthcare supply chains functions as a distributed trust infrastructure that ensures product authenticity, regulatory traceability, cold chain integrity, and multi-party transparency. Unlike traditional centralized databases, blockchain operates as a shared, immutable ledger where all authorized stakeholders validate and record transactions.
In life sciences, where product safety directly impacts patient survival, this architecture becomes strategically critical.
1️⃣ Why Blockchain Is Critical in Pharma & Life Sciences
The pharmaceutical supply chain is highly fragmented:
- API manufacturers
- Drug manufacturers
- Contract manufacturing organizations (CMOs)
- Distributors & wholesalers
- Logistics providers
- Hospitals & pharmacies
- Regulators
Each transfer increases the risk of:
- Counterfeit drug insertion
- Product diversion
- Temperature excursions
- Documentation fraud
- Delayed recalls
The World Health Organization estimates that counterfeit medicines account for up to 10% of the global supply in certain regions. This creates patient safety risks and billions in revenue losses.
Blockchain addresses these structural inefficiencies by creating end-to-end product traceability.
2️⃣ Core Technical Architecture
Most pharma implementations use permissioned blockchain networks, not public cryptocurrency systems.
Key Components
A. Distributed Ledger
Each transaction is:
- Time-stamped
- Cryptographically secured
- Immutable
- Replicated across nodes
B. Nodes
Every major stakeholder (manufacturer, distributor, regulator) operates a node that validates transactions.
C. Smart Contracts
Automated rules embedded in blockchain logic.
D. IoT Integration
Temperature sensors and GPS trackers feed real-time data into the ledger.
Enterprise platforms frequently leverage frameworks like IBM blockchain services for healthcare networks.
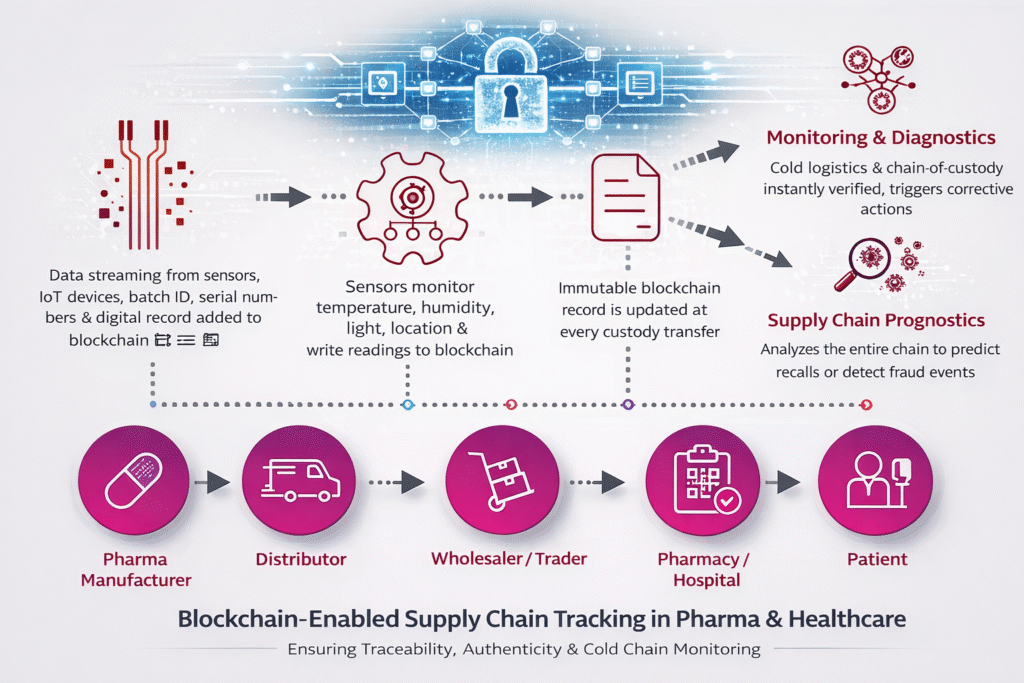
3️⃣ Step-by-Step Workflow in Pharmaceutical Supply Chain
Step 1: Manufacturing & Batch Creation
When a pharmaceutical company such as Pfizer produces a drug batch:
- Batch ID
- Lot number
- Manufacturing timestamp
- Active ingredient source
- Compliance certification
- Expiry date
These data points are written as the first block in the chain.
This creates a digital birth certificate for the drug.
Step 2: Serialization & Unit-Level Identification
Regulations such as the U.S. Drug Supply Chain Security Act (DSCSA) require unit-level serialization.
Each unit receives:
- Unique serial number
- QR code / RFID
- Linked blockchain identity
This prevents duplication and clonin
Step 3: Logistics & Cold Chain Monitoring
Biologics, vaccines, and gene therapies require strict temperature ranges (2–8°C or ultra-cold).
IoT sensors record:
- Temperature
- Humidity
- Location
- Transit time
If temperature exceeds threshold:
Smart contract triggers alert.
Companies including Moderna have explored blockchain-enabled vaccine tracking to ensure cold chain compliance.
Step 4: Distributor & Wholesaler Transfer
Each custody transfer is logged:
Manufacturer → Distributor → Wholesaler → Pharmacy → Hospital
Blockchain records:
- Time of transfer
- Receiving party ID
- Condition verification
- Shipping container ID
This ensures chain-of-custody transparency.
Step 5: Pharmacy & Hospital Authentication
Before dispensing:
- QR code scanned
- Blockchain verifies authenticity
- Confirms complete transaction history
Counterfeit or diverted products are immediately flagged.
4️⃣ Use Cases Across Healthcare & Life Sciences
A. Anti-Counterfeit Control
Blockchain prevents fake drugs from entering legitimate supply channels by:
- Serial verification
- Duplicate detection
- Real-time validation
B. Faster & Targeted Drug Recalls
Traditional recall process:
Recall entire distribution network.
Blockchain-enabled recall:
Identify affected batch → Trace exact downstream locations → Notify only impacted facilities.
This reduces financial and reputational damage.
C. Clinical Trial Supply Management
Clinical trials involve:
- Investigational drugs
- Multiple global trial sites
- Strict regulatory documentation
Blockchain ensures:
D. Medical Device Tracking
Implants and surgical devices can be serialized and tracked, preventing counterfeit or expired devices from entering operating rooms.
E. Genomic & Research Data Integrity
Life sciences research institutions collaborate globally.
Blockchain enables:
- Secure genomic data sharing
- Data access audit trails
- Consent tracking
5️⃣ Measurable Operational Benefits
1. Reduced Fraud Losses
Prevents counterfeit drug infiltration.
2. Improved Regulatory Compliance
Real-time auditability for regulators.
3. Lower Recall Costs
Precision recall instead of blanket recall.
4. Operational Transparency
Eliminates manual reconciliation between parties.
5. Enhanced Patient Safety
Authenticity verification at dispensing point.
6️⃣ Economic Impact & Market Outlook
Blockchain in healthcare supply chains is projected to grow due to:
- DSCSA full enforcement deadlines
- Increased biologics and mRNA therapies
- Global vaccine distribution complexities
- Rising cross-border pharmaceutical trade
As drug modalities become more temperature-sensitive and personalized (e.g., cell & gene therapy), traceability complexity increases — making blockchain more relevant.
7️⃣ Challenges & Implementation Barriers
Integration Complexity
ERP, EHR, warehouse systems must integrate with blockchain nodes.
High Initial Costs
Infrastructure, onboarding, compliance mapping.
Stakeholder Alignment
Blockchain only works if all major participants join.
Data Privacy Regulations
HIPAA, GDPR compliance must be maintained.
Blockchain is not a standalone fix; it requires ecosystem coordination.
8️⃣ Blockchain + AI + IoT Convergence
Future supply chain architecture will combine:
Blockchain → Data trust
IoT → Real-time environmental data
AI → Predictive logistics & anomaly detection
For example:
AI predicts delay risk → Smart contract adjusts shipping route → Blockchain records changes transparently.
This creates a self-optimizing pharmaceutical supply network.
9️⃣ Strategic Implications for 2026–2030
Pharma companies adopting blockchain early will gain:
- Stronger compliance posture
- Competitive differentiation
- Lower counterfeit exposure
- Greater supply chain resilience
Governments are increasingly mandating serialization and transparency, which accelerates blockchain adoption.
Conclusion
Blockchain in pharma, healthcare, and life sciences is fundamentally about establishing distributed trust across complex ecosystems. It transforms opaque supply chains into transparent, verifiable networks where every transaction is authenticated, recorded, and auditable.
While challenges remain in integration and cost, regulatory pressure and counterfeit risk are driving enterprise adoption. As biologics, gene therapies, and personalized medicine expand, blockchain will become a core infrastructure layer — not just an optional enhancement — in global healthcare supply chains.
FAQ – Blockchain in Pharma & Healthcare Supply Chain
1. What is blockchain in pharmaceutical supply chain?
Blockchain is a distributed, tamper-proof ledger that records every transaction in the drug supply chain—from manufacturing to pharmacy dispensing—ensuring traceability and authenticity.
2. How does blockchain prevent counterfeit drugs?
Each drug unit receives a unique serialized ID linked to a blockchain record. When scanned at hospitals or pharmacies, the system verifies its full transaction history, preventing fake or duplicated products from entering the system.
3. How is blockchain used in cold chain monitoring?
IoT sensors track temperature, humidity, and transit conditions. This data is written to blockchain, creating an immutable environmental log. Smart contracts can trigger alerts if temperature thresholds are violated.
4. Is blockchain compliant with pharmaceutical regulations?
Yes, blockchain supports compliance with serialization and traceability regulations such as the U.S. DSCSA and EU FMD by providing real-time, auditable transaction records.
5. What are smart contracts in healthcare supply chains?
Smart contracts are automated rules stored on the blockchain. For example, if a shipment exceeds temperature limits, the contract can automatically flag the batch or notify stakeholders.
6. Which companies are implementing blockchain in pharma?
Technology providers like IBM and pharmaceutical companies such as Pfizer and Moderna have explored blockchain for vaccine tracking and supply chain transparency.
7. What are the main benefits of blockchain in life sciences?
Key benefits include improved transparency, reduced fraud, faster recalls, enhanced regulatory compliance, and increased patient safety.
8. What are the challenges of implementing blockchain in healthcare supply chains?
Major challenges include high integration costs, interoperability between systems, stakeholder coordination, and maintaining data privacy under regulations like HIPAA and GDPR.
9. Can blockchain integrate with AI and IoT in pharma?
Yes. IoT devices collect real-time logistics data, blockchain ensures data integrity, and AI analyzes patterns to predict disruptions—creating a resilient, intelligent supply network.
10. What is the future of blockchain in healthcare supply chains?
Between 2026 and 2030, blockchain is expected to become a core infrastructure layer supporting biologics, gene therapies, precision medicine logistics, and global regulatory compliance systems.