1. Introduction to 3D Printing of Medication
3D printing of medication, also known as pharmaceutical additive manufacturing, represents a paradigm shift in drug formulation and delivery science. Unlike conventional pharmaceutical manufacturing—which relies on batch processing, compression, granulation, and coating—3D printing constructs drug products layer by layer using a digital design file. This manufacturing flexibility enables unprecedented control over drug geometry, internal porosity, dosage accuracy, and release kinetics.
Traditional drug manufacturing follows a “one-dose-fits-all” approach. Patients often receive standardized dosages that may not perfectly match their metabolic rate, age, genetic profile, or disease severity. In contrast, 3D printing enables precision medicine by allowing pharmacists or manufacturers to produce patient-specific tablets tailored to individual therapeutic needs.
The technology integrates pharmaceutical sciences, materials engineering, computational modeling, and biomedical innovation. By combining drug substances with printable polymers or excipients, customized dosage forms can be designed to dissolve instantly, release slowly over time, or deliver multiple drugs in a structured sequence.
The first FDA-approved 3D-printed drug, Spritam, marked a milestone in pharmaceutical history. This approval demonstrated regulatory feasibility and opened doors for broader adoption.
As healthcare shifts toward personalized therapy, decentralized manufacturing, and AI-driven formulation modeling, 3D printing of medication stands at the intersection of innovation and patient-centric care.
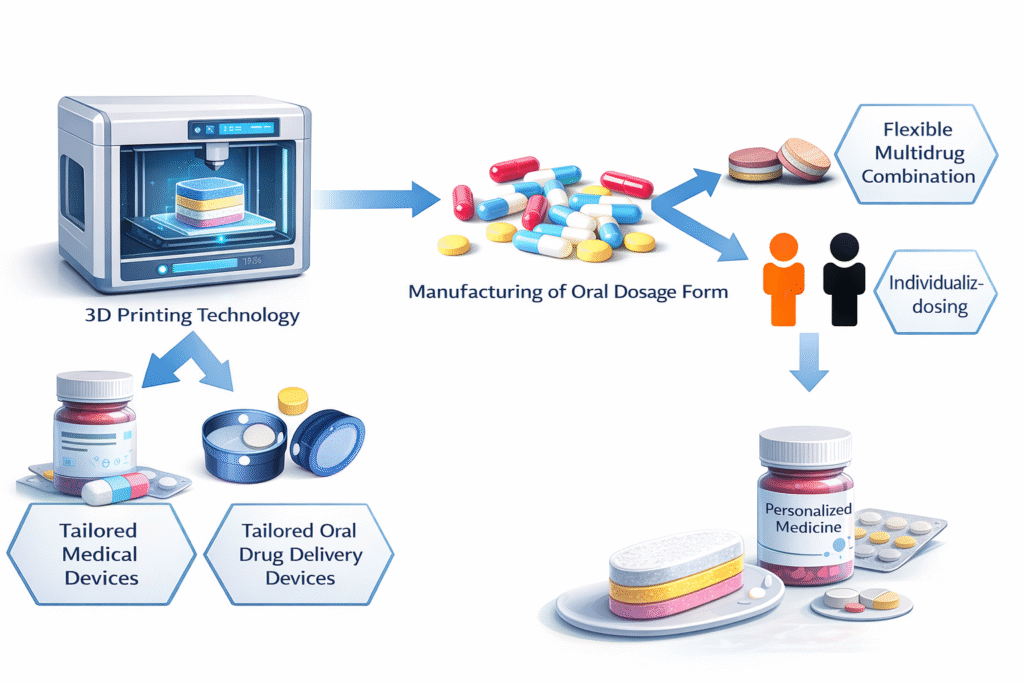
2. How 3D Printing of Medication Works
The 3D printing process in pharmaceuticals begins with digital formulation design. Scientists create a computer-aided design (CAD) model specifying tablet shape, dose, porosity, and internal structure. Advanced modeling tools allow simulation of drug diffusion rates and dissolution behavior before physical production begins.
The next stage involves material preparation. Active pharmaceutical ingredients (APIs) are blended with excipients such as polymers, plasticizers, and binders to create printable feedstock. Depending on the technology used, this material may take the form of powder, filament, paste, or liquid ink.
During printing, the machine deposits or fuses material layer by layer following the digital blueprint. This precise layering allows micro-architectural control over drug distribution inside the dosage form. For example, a tablet can contain multiple compartments, each programmed to release medication at different times.
Post-processing steps such as drying, UV curing, or coating ensure product stability and compliance with pharmacopoeial standards.
This digitally controlled workflow reduces waste, enables rapid prototyping, and allows on-demand production—making it highly suitable for hospital pharmacies and specialized treatment centers.
3. Major 3D Printing Technologies Used in Pharmaceuticals
Several additive manufacturing techniques are adapted for pharmaceutical use:
Binder Jetting (Inkjet Printing) – A liquid binder containing the drug is sprayed onto powder layers. This method is ideal for highly porous, fast-dissolving tablets.
Fused Deposition Modeling (FDM) – Drug-loaded polymer filaments are melted and extruded through a heated nozzle. Suitable for sustained or controlled-release formulations.
Semi-Solid Extrusion (SSE) – Gel or paste-based drug mixtures are extruded at low temperatures, making it suitable for heat-sensitive APIs.
Selective Laser Sintering (SLS) – A laser selectively fuses powder particles, enabling complex internal drug structures.
Each technique offers unique advantages in dose flexibility, structural precision, and drug stability.
4. Advantages of 3D Printed Medication
One of the most significant benefits is personalized dosing. Patients with chronic diseases often require dose adjustments based on metabolism, weight, or genetic factors. 3D printing eliminates the need to split tablets or combine multiple prescriptions manually.
The polypill concept is another breakthrough. Multiple drugs can be incorporated into a single tablet with distinct release compartments. This improves adherence, especially among elderly patients managing several medications.
Complex release profiles—immediate, delayed, pulsatile, or sustained—can be engineered into one dosage form. This reduces dosing frequency and enhances therapeutic effectiveness.
On-demand manufacturing also minimizes storage needs and drug wastage, making supply chains more efficient.
5. Challenges and Regulatory Considerations
Despite its promise, several barriers remain. Regulatory frameworks are still evolving to address quality assurance, reproducibility, and batch validation for personalized production.
Material stability is another challenge. Some drugs degrade under heat during extrusion-based printing.
Scalability for mass production remains under investigation. While 3D printing excels in customization, traditional methods may remain more economical for high-volume generics.
However, as regulatory agencies refine guidelines and technology advances, these limitations are expected to diminish.
6. Applications in Healthcare and Life Sciences
3D printed medication is particularly valuable in:
- Pediatric care (custom dose sizes and shapes)
- Geriatric medicine (easy-to-swallow porous tablets)
- Oncology (precision chemotherapy dosing)
- Rare diseases (low-volume customized therapies)
- Hospital-based emergency drug production
Integration with AI-driven modeling and digital twin simulations can further optimize drug design for individual patient physiology.
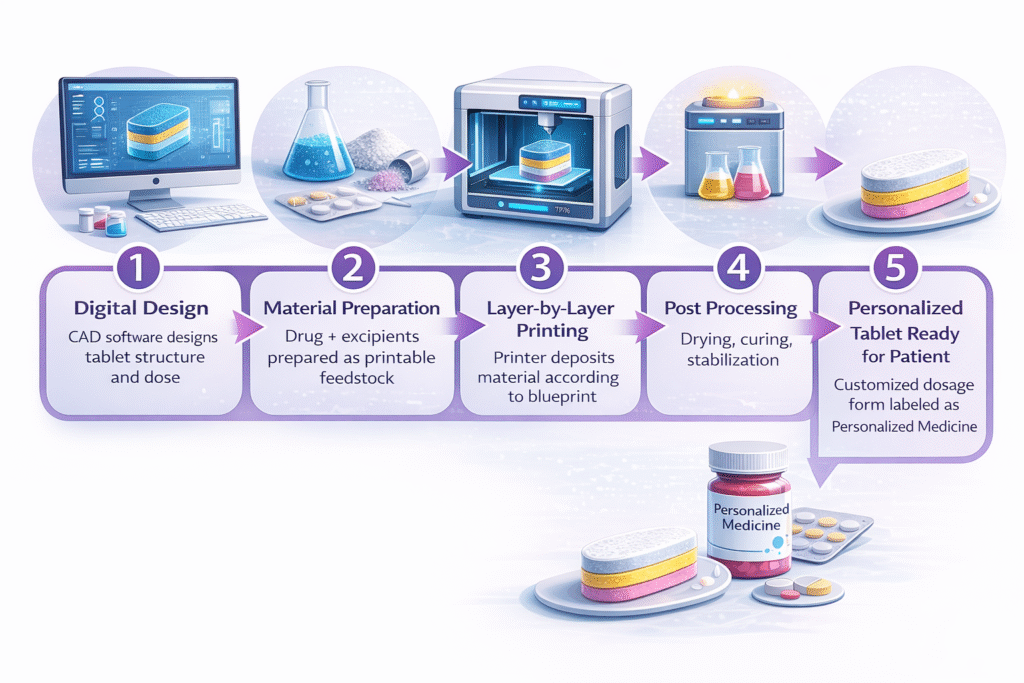
Step 1: Digital Design
→ CAD software designs tablet structure and dose
Step 2: Material Preparation
→ Drug + excipients prepared as printable feedstock
Step 3: Layer-by-Layer Printing
→ Printer deposits material according to blueprint
Step 4: Post Processing
→ Drying, curing, stabilization
Step 5: Personalized Tablet Ready for Patient.
FAQ Section
1. What is 3D printing of medication?
It is an additive manufacturing process that produces drug tablets layer by layer using a digital model, enabling personalized dosing and controlled release.
2. Is any 3D printed drug approved?
Yes. Spritam was approved for epilepsy treatment.
3. What are polypills?
Polypills combine multiple medications into a single tablet using compartmentalized 3D structures.
4. Is 3D printed medication safe?
When manufactured under regulatory standards and validated processes, it meets pharmaceutical safety requirements.
5. Can hospitals print medicines on demand?
Yes, future hospital pharmacies may adopt on-site drug printing for personalized therapy.
6. What industries benefit most?
Pharmaceutical manufacturing, biotechnology, personalized medicine, and hospital healthcare systems.
